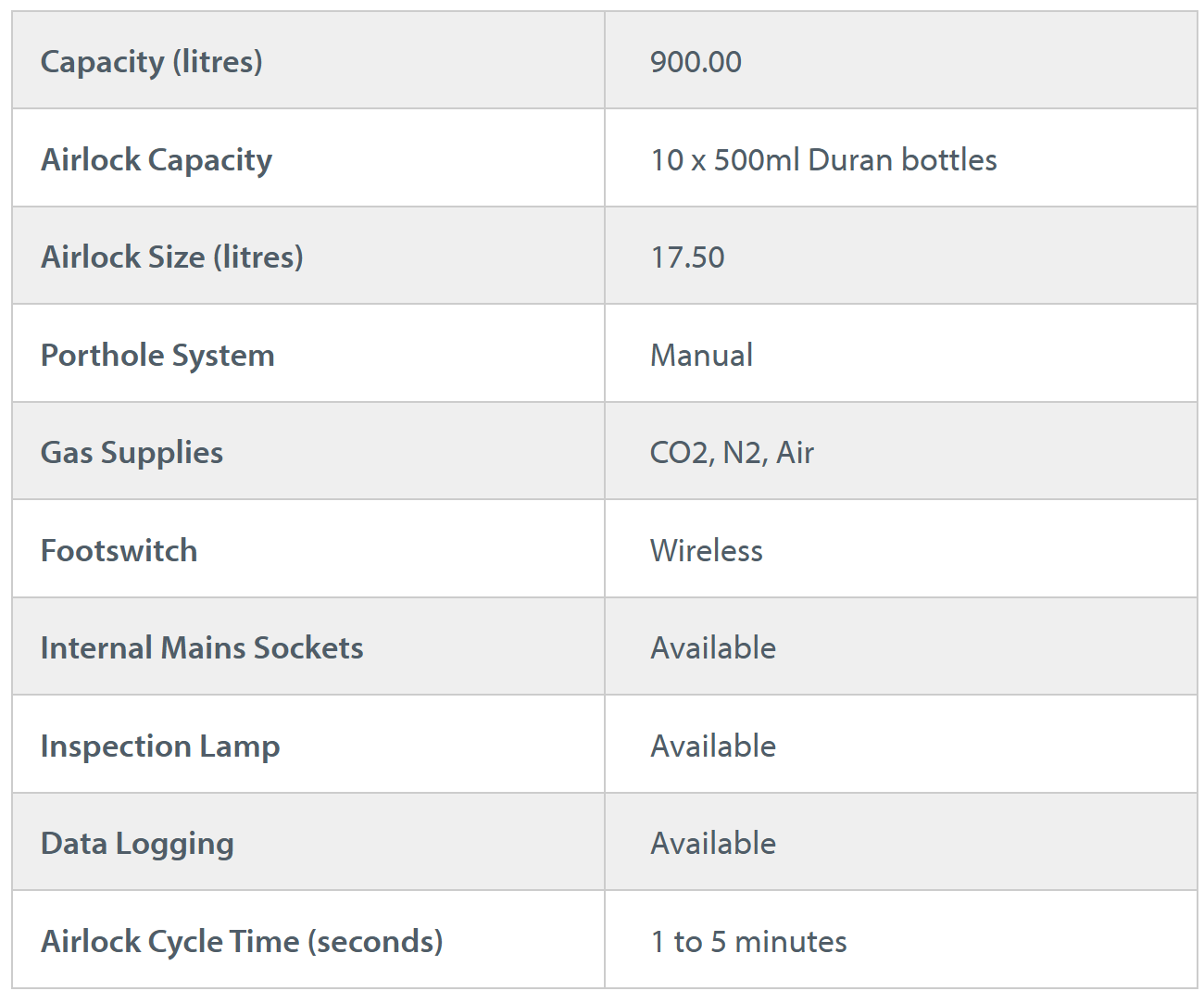
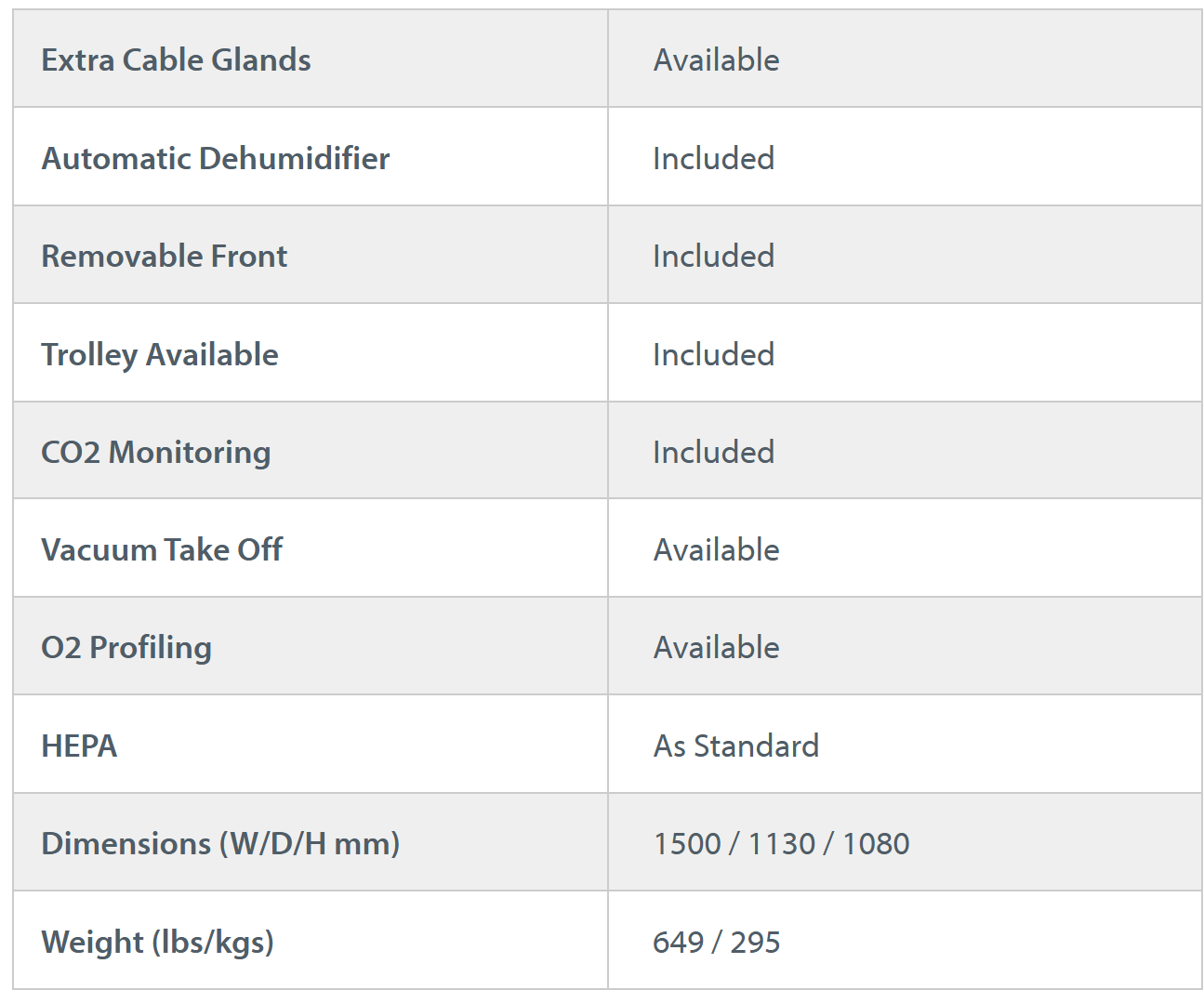
Whitley H135 GMP Isolator
Cell Optimised Isolator providing Grade A laminar airflow and low oxygen control for stem cell products
The Whitley H135 GMP Isolator (GB Patent No. 2593277) is designed to fast track your allogenic approval. Experience unrivalled control and consistency through enhanced contamination reduction, cell behaviour control, and cell growth.
The Whitley Difference:
Contamination Control: ISO3 unidirectional flow - 100X less particles than Grade A. Crucial for safe and effective ATMPs.
Control & Consistency: Ensure consistent processing with customisable atmosphere control (oxygen, carbon dioxide, humidity, and temperature) for in vivo, low oxygen conditions. Improving control of cell behaviour, proliferation, and differentiation.
Enhanced Cell Growth: large incubation and processing capacity with atmosphere control for increased stem cell production and yields.
Designed to ensure batch-batch consistency for:
- GMP Allogenic Manufacturing.
- Regenerative Medicine.
- iPSC In Vitro Models.
- Tissue Engineering.
- Organoids.
- QC Potency and Efficacy Testing.
- Tumour Microenvironment Assays.
Utilising low oxygen control with ISO 3 (100X less particles than Grade A) laminar flow creating an in vivo environment for optimised stem cell growth.
With a compact and closed design, our re-circulating technology enables our isolators to be housed in Grade D cleanrooms. Helping you develop scalable and cost-effective cleanroom alternatives capable of saving $20,000/m in operations.
Process, incubate and refrigerate your oxygen sensitive products in Grade A conditions without the need to expose cells to atmospheric conditions. Avoiding HIF-1 oxygen shock and contamination risks associated with incubator-BSC transfer.
Gauntlets
GMP regulations require specialised robust gauntlets to be used in GMP high risk processing applications. Our Cell Optimised Isolators are supplied with an innovative quick change ‘BCS’ gauntlet made of non-shedding material allowing easy and rapid glove changes without loss of containment or atmospheric conditions.
MODULARITY
Incubation Module
- Relative humidity control up to 80%.
- Incubation at 37°C to support cell growth.
- CO₂ control with 0.1% increments between 0.1% - 15% CO₂ in all modules.
- O₂ control with 0.1% increments between 0.1% - 20% O₂ in all modules for hypoxic stem cell culturing.
Cell Processing Module
- Equipment integration for seamless operation within the isolator.
- Aseptic transfer system to facilitate use of external equipment inside the isolator.
- CO₂ control with 0.1% increments between 0.1% - 15% CO₂ in all modules.
- O₂ control with 0.1% increments between 0.1% - 20% O₂ in all modules for hypoxic stem cell culturing.
Refrigeration Module
- Relative humidity control.
- Grade A refrigeration for cell culture reagents at 7°C.
- CO₂ control with 0.1% increments between 0.1% - 15% CO₂ in all modules.
- O₂ control with 0.1% increments between 0.1% - 20%.

- Precise oxygen control (between 0.1% and 20%) and CO₂ control (between 0.0% and 15%).
- Precise control of temperature (from ambient to incubation temperature) and humidity.
- The standard H14 HEPA filter is better than 99.995% efficient at the most penetrating particle size (MPPS), and the upgraded U15 ULPA filter is better than 99.99995% efficient at the MPPS.
- DWS HEPA and ULPA filters are packed with 33 pleats per decimetre to ensure we achieve the required downflow rate.
- Large work chamber accommodates a variety of imaging, centrifuge and cell processing equipment for a custom setup.
- Ethernet-enabled for remote access review and control.
- Whitley Intelligent Monitoring - Environmental and atmospheric sensors measuring and controlling airflow, airspeed, positive pressure, O₂, CO₂, temperature, and humidity.
- Data logging system records parameters every 60 seconds.
- Intuitive HMI with individual user logins and levels.
- Glove and leak testing.
- Sterile liquid transfer ports.
- Equipment integration with power sockets.
- Event Log capturing all user, event and alarm records ensuring data integrity.
- External EMS and sensor connections.
- Data Logging with Ethernet download to servers.
- Remote ethernet access.
- Full URS, FAT, SAT, IQ/OQ, RQ validation packages.
- 'Standby' feature (Eco mode) automatically decreases fan speed when the chamber is at rest to optimise energy efficiency.
- Continuous viable and non-viable particle monitoring.
- Active air sampling.
- Fully automated dehumidification system.
- Bespoke trolley with cleanroom castors.
- Other optional features are available to tailor the system to your requirements.